  Carboxylic Acid Questions 1. 
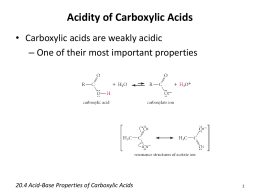
If you have any queries, drop a comment below, and we will get back to you. View Carboxylic Acid Questions-1.doc from CO 2H at Harvard University. stabilization in carboxylates makes carboxylic acids more acidic than. We hope this article on the Derivatives of Carboxylic Acids has helped you. Arrange the following in order of (i) increasing acidity, (ii) increasing pKa. Answer : Br is an electron attracting group.Electron attracting groups (I groups) increase the acidic strength of carboxylic acids. The correct order of acid strength of the following carboxylic acids is JEE. can read more about the inductive effect and the pKa of carboxylic acids in. Among them, the carboxylic acid is a functional group-containing carboxyl group represented as \(.\) It first reduces the carboxylic acid to aldehyde, which rapidly reduces it to alcohol. Arrange the following carboxylic acids inorder of decreasing acid strength. Question : Arrange the following carboxylic acids in increasing order of their acid strength: CH 3 CH 2 CH(Br)COOH, CH 3 CH(Br)CH 2 COOH, (CH 3) 2 CHCOOH, CH 3 CH 2 CH 2 COOH. Arrange the following in the increasing order of their acidic strength. Example: Rank the following compounds in order of increasing acidity without. In organic chemistry, there are various functional groups. From 1 being the least acidic and 4 being the most acidic, arrange the following organic compounds in terms of increasing acidity. aldehydes ketones carboxylic acids class-12 1 Answer 1 vote answered by RakshitKumar (35.7k points) selected by PulkitKumar Best answer Acidity in the decreasing order. Arrange the following carboxylic acids with increasing order of their acidic strength and justify your answer. #ARRANGE THE FOLLOWING CARBOXYLIC ACIDS IN ORDER OF ACIDITY TRIAL#Start your trial now First week only 6.99 arrow. Arrange the following acids in order of their decreasing acidity. Whatever be the composition of the rest of the molecule, the same functional group in the molecules will undergo similar chemical reactions. Solution for Arrange the following types in order of increasing acidity: carboxylic acids, alcohols, phenols, water. Derivatives of Carboxylic Acids: A substituent or moiety in a molecule that is responsible for the molecule’s characteristic chemical reactions is known as a functional group. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
